|
Positions are measured using the meter sticks, then wavelengths are determined from the positions using the graph itself or the equation of the best fit line for that graph.įor atoms that contain only one electron, the theory of atomic structure proposed by Niels Bohr can be used to calculate wavelengths for transitions between particular electronic energy levels of the atom. The calibration graph is therefore an integral part of the spectroscope. Gaussian fit to the data (black dotted line). Inset: magnified peak of the emission line at 546.0735 nm (literature value, entrance slit approx. Data points (blue), literature peak positions of atomic emissions (red). For example, using the same apparatus and without moving the relative positions of the meter sticks, diffraction grating and lamp, it is possible to view the spectrum of a new element, measure where its spectral lines occur on the meter stick, and then read the graph or use the equation of the line to determine the wavelength to which each of those positions corresponds. Atomic emission spectrum of a mercury vapour lamp as measured with the DIY CzernyTurner spectrometer. 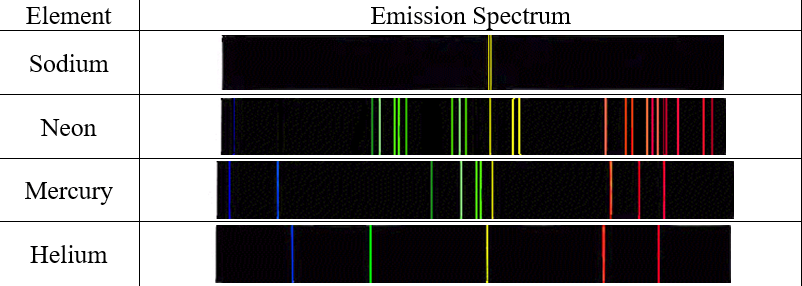
Once the best fit straight line has been determined, the equation of this line can then be used to convert positions of other spectral lines to wavelength. position of the spectral line will yield a straight line. Since this position depends upon the wavelength in a linear way, a graph of wavelength vs. Using a light source that contains known wavelengths of light, we can measure exactly where each known wavelength appears along a meter stick. As the light emerges after being reflected by the grating, these tiny lines cause the reflected light to interfere with itself in such a way that the different wavelengths of the light to appear in different positions to the left and right of the original direction in which the light was traveling. A diffraction grating is a piece of glass or clear plastic with many very narrow and closely spaced lines on it. If we view the light through a prism or a diffraction grating, however, the individual wavelengths are separated. To the naked eye, the various wavelengths (colors) of light emitted by an element are mixed together and appear as a single color that is a combination of the component colors. 
To measure these wavelengths in the laboratory, we must first separate them. Thus, the spectrum of an element can be stated by listing the particular wavelengths of light that its atoms emit. These two relationships combine to give a third: I would encourage you to explore further.Js\). looking for relationships in a meaningful way. We can, however, say that the more electrons there are in an atom, the greater the variety of photon energies there will be so this may be a link worth exploringĪs I say, you are asking a great question. as it switches from one energy level to another, then it emits / absorbs photons. This is because the electron can exist in many energy levels. Hydrogen, for example, although the simplest atom, has a whole range of photon energies that it emits. The value of the energies of photons does depend on the available energy levels in the atom.  
Now, the energy of the photon emitted from any element does not depend on the number of electrons in the atom. OK: I would say that the periodic table tells us about the number of protons in an element and, therefore the number of electrons too. Here is my answer, but I would encourage you to explore this and similar questions further. Planck studied the electromagnetic radiation emitted by heated objects, and he proposed that the emitted electromagnetic radiation was "quantized" since the energy of light could only have values given by the following equation: E photon = n h ν E_ s 1 start fraction, 1, divided by, start text, s, end text, end fraction. Physicists Max Planck and Albert Einstein had recently theorized that electromagnetic radiation not only behaves like a wave, but also sometimes like particles called photons. By the early 1900s, scientists were aware that some phenomena occurred in a discrete, as opposed to continuous, manner.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
